文章信息
- 隋凤凤, 王静波, 吴昊, 李恋卿, 潘根兴
- SUI Feng-feng, WANG Jing-bo, WU hao, LI Lian-qing, PAN Gen-xing
- 生物质炭钝化农田土壤镉的若干研究进展
- Several research progresses in Cd inactivation by biochar application in agricultural soil
- 农业环境科学学报, 2018, 37(7): 1468-1474
- Journal of Agro-Environment Science, 2018, 37(7): 1468-1474
- http://dx.doi.org/10.11654/jaes.2018-0667
文章历史
- 收稿日期: 2018-05-21
- 录用日期: 2018-06-11
随着工业化和城市化的迅猛发展,环境中的重金属污染情况日益严重[1]。采矿、冶炼以及农业生产中磷肥、污泥的施用等活动均加剧了土壤重金属的污染[2-3]。镉在土壤中具有较强的蓄积性、迁移能力和生物可积累性,是最有毒性的无机污染物之一[4-5]。镉主要通过食物摄入的方式进入人体,在人体中的半衰期长达20~40年,对骨骼及肾脏造成严重危害。联合国环境规划署将镉列在优先污染物名单中的首位[6]。据2014年原环保部的数据显示,我国土壤镉污染的点位超标率达7%,远高于汞(1.6%)、砷(2.7%)、铅(1.5%)等其他重金属元素[7]。近年来,由于土壤镉污染造成的粮食作物的镉超标现象引起了极大的关注。Liu等[8]的研究结果表明,我国稻田的镉污染浓度范围为0.01~5.5 mg·kg-1。其中,南方重金属污染稻田镉污染相对严重,70%的稻米存在镉污染,对我国部分市售大米的抽样检测结果显示,镉超标率达10%[9-10]。针对日益严重的土壤镉污染问题,采取有效的治理措施以保障粮食安全生产一直是相关领域的研究热点。
生物质炭是限氧条件下热裂解生物质获得的一种固体材料,含有稳定态有机碳以及可溶性有机及矿质灰分等多组分混合物,生物质炭可明显提高土壤质量和生产力。研究显示,施用生物质炭可改善土壤结构、土壤持水量、提高土壤有机质含量、促进土壤养分循环并提高作物产量[11-17]。同时,生物质炭由于具有高pH、高比表面积、丰富的孔隙及表面官能团等特性,作为重金属污染土壤的修复材料受到环境修复界的广泛关注[18-21]。本文试图总结近十多年来生物质炭对土壤镉钝化并抑制植物吸收的研究进展,期望为农业土壤镉污染治理及污染土壤修复和未来研究问题提供科学参考。
1 生物质炭抑制土壤-作物系统镉迁移转化:多作用机制大量研究证实,生物质炭对镉在土壤-作物系统中的运移具有较好的钝化效果[20-25]。据广西、湖南、江西等多点野外试验结果表明,施用20~40 t·hm-2的小麦秸秆炭,土壤有效态镉含量降低20%~70%,水稻籽粒镉积累量降低20%~60%[22-24]。Chen等[25]通过整合分析显示,生物质炭处理对土壤有效态镉的平均降幅为52%。Liu等[26]发现施用20 t·hm-2的水稻秸秆和豆秸炭,稻田田面水中的可溶性镉含量分别降低56%~80%和61%~83%。另一研究表明,生物质炭在1%、2%以及4%的施用量下,土壤可交换态镉降低了28.5%~59.4%,对水稻镉积累量的降幅为2.7%~23.8%[25]。类似地,在1.5%、3%以及5%的施用范围内,生物质炭对小麦籽粒镉的降低幅度可分别达到26%、42%以及57%[28]。由此可见,生物质炭在降低土壤镉的生物有效性和抑制作物镉积累方面具有明显的效果。
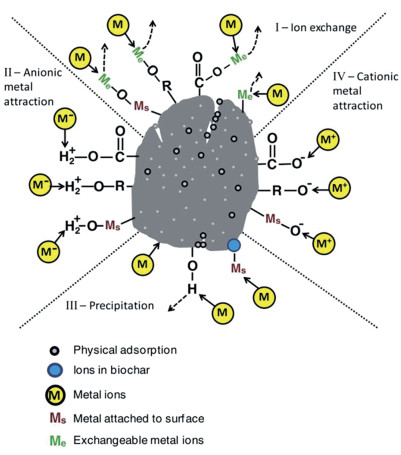
根据Ahmad等[29]的研究发现(图 1),并结合目前相关研究结果,生物质炭对土壤中重金属离子可能的作用机制大致可表现为以下几个方面:(1)生物质炭较大的表面积使得镉可以通过与生物质炭中的K+与Na+等有效态阳离子的交换作用而结合于生物质炭的表面;(2)镉与生物质炭表面官能团相互作用产生的表面络合;(3)生物质炭中较高含量的灰分元素可通过增加土壤pH以及碳酸盐、磷酸盐等无机盐含量而与镉产生沉淀或络合作用[29-33]。
首先,生物质炭由于含有较多的可交换态阳离子以及较大的表面积,可通过离子交换作用将镉固持于表面[21, 24]。研究表明,生物质炭表面的H+、Ca2+以及K+等可交换态阳离子对镉生物有效性的降低具有重要作用[34-36]。Zhang等[37]研究发现,藻类生物质炭释放出的K+、Ca2+、Na+、Mg2+等阳离子的总量几乎与所固持的镉的总当量相等。Harvey等[38]比较松木和灌木生物质炭发现,具有较高CEC的木材生物质炭对镉的吸附机制主要以离子交换作用为主。此外,Xu等[39]发现镉、锌、铅、铜会竞争稻壳炭中质子化的酚羟基官能团结合位点。Mohamed等[40]研究表明,施用1.5%的竹炭后,土壤中DTPA浸提态镉含量从33.32 mg·kg-1降低至12.09 mg·kg-1,这可能是生物质炭表面官能团与土壤中的镉发生了络合作用从而降低了镉的生物有效性[29]。
其次,生物质炭灰分中可溶性盐、可溶性有机成分的溶解以及炭表面官能团的解离均能够增加土壤pH [41-45],这也是生物质炭降低土壤镉生物有效性的重要机制之一[38]。Rees等[46]研究显示,生物质炭能够同时增加酸性和碱性土壤的pH。同时,生物质炭中的矿质组分可通过与镉产生的共沉淀及内部络合作用降低镉的生物有效性[19-20]。Trakal等[47]通过FTIR图谱发现葡萄秸秆炭以及稻壳炭对镉的吸附会引起CO32-峰的改变,这表明镉可能与CO32-在生物质炭表面形成了共沉淀。Zhang等[37]利用X射线衍射仪发现,藻类生物质炭对镉的吸附伴随着碳酸镉及磷酸镉矿物的形成。Xu等[39]的研究表明低温畜禽粪便炭(350 ℃)中磷酸根及碳酸根对镉的吸附贡献分别为 < 10%及 > 90%,而通过酚羟基的结合产生的表面吸附所占比例 < 25%。此外,生物质炭对土壤pH的增加还可增加土壤胶体表面的负电荷,从而进一步降低重金属的运移风险[48]。多年定位试验发现,生物质炭对土壤pH的增幅会逐年降低,但对镉的钝化效应却具有一定的持效性。这可能是由于土壤的生物及非生物氧化作用增加了老化生物质炭表面的含氧官能团,进而促进了生物质炭对镉的吸附作用[41-43]。Bian等[49]的田间试验证实,田间老化三年的生物质炭颗粒表面的含氧官能团有所增加。
生物质炭抑制作物镉吸收的效应与土壤中镉的钝化有直接关系[38, 45-47]。研究显示,生物质炭处理下水稻籽粒镉的降低量与土壤中CaCl2浸提态镉的降低量存在显著正相关关系[50-51]。Xu等[52]的研究结果显示,5%(W/W)的花生壳炭能够显著降低土壤中MgCl2浸提态镉的含量,对水稻籽粒镉含量的降低幅度达22.9%。此外,生物质炭还可通过元素的相互作用来抑制作物对镉的吸收。整合分析结果显示,生物质炭对镉在土壤-作物体系中运移的抑制效果均高于锌[11, 25]。通常情况下,施用锌肥可抑制作物对镉的吸收[53-55]。施用含锌量高的生物质炭(如畜禽粪便炭)可提高作物对锌的吸收从而降低镉的积累[11, 56]。此外,水稻秸秆类生物质炭还可增加土壤中有效态的硅含量,而硅可通过在水稻和小麦的根部内皮层形成沉淀来阻隔镉从根部向地上部的运移[57-58]。因此,施用含硅量较高的生物质炭可进一步抑制作物对镉的吸收和积累。
2 生物质炭的土壤镉钝化效应:多因素影响 2.1 原料和热裂解温度生物质炭的性质是由原料和制备工艺共同决定的。不同原料及工艺制备的生物质炭对重金属的钝化效应存在一定的差异[41, 59]。与秸秆、木材等原料相比,畜禽粪便类生物质炭对镉的钝化效果相对较好,这是由于畜禽粪便炭的灰分含量及阳离子交换量较高,可与土壤中的镉形成共沉淀或者通过离子交换作用抑制镉的运移[29, 41, 60-62]。Xu等[39]研究显示牛粪炭对镉、锌、铅、铜的作用主要以碳酸根以及磷酸根的沉淀作用为主。
热裂解温度也是影响生物质炭对镉钝化效果的重要因素。在一定范围内,生物质炭的灰分含量随着热裂解温度的增加而增加,pH也随之增加[29]。Xu等[39]的研究显示当热裂解温度从200 ℃升高至350 ℃时,生物质炭的pH由7.63上升到9.98,对镉的吸附量从31.9 mg·g-1提高到51.4 mg·g-1,其中可溶性CO32-组分具有重要贡献。此外,生物质炭的挥发分、含氧组分以及电荷零点等均能影响其表面官能团的形成[29],较高热裂解温度(> 600℃)制备的生物质炭会出现CEC下降、挥发性组分损失、比表面积增加以及表面含氧官能团数量和电荷密度下降等现象,生物质炭表面官能团数量也随之下降[63]。因此,从生物质炭的结构特性来看,在200~450 ℃的热裂解温度范围内制备的生物质炭具有相对丰富的含氧官能团、较高的静电引力以及较高的可溶性无机盐含量,从而对土壤中的镉具有较好的钝化效果[11, 29]。
2.2 土壤pH和有机质土壤的pH和有机质等性质可通过影响土壤的酸碱性、吸附性以及氧化还原状态而对生物质炭的钝化效果产生一定的影响[64-71]。通常, 实际应用的生物质炭大多呈碱性[72],这是由于生物质炭对土壤pH的增加可以抑制重金属在土壤中的运移[50, 73-74]。当石灰效应较弱时,生物质炭对镉可能无显著的钝化效果。Qi等[75]的研究发现,酸性刨木炭(pH=3.25)对偏酸性(pH=6.14)及偏中性(pH=7.87)土壤中的镉均无明显钝化作用。另一盆栽试验结果显示,5%(W/W)生物质炭(pH=9.46)的施用增加了碱性土壤(pH=8.0)中超积累作物天蓝遏蓝菜(Noccaea caerulescens)对镉的积累量[76]。在长期定位试验中,碱性生物质炭在酸性土壤中对镉的钝化效果比在碱性土壤明显。Bian等[77]在太湖地区进行的大田(pH=5.36)试验结果表明,小麦秸秆炭(pH=10.4)能够连续三年显著增加土壤的pH,同时显著降低土壤可提取态镉含量及水稻籽粒镉积累量。而Sui等[78]在我国北方碱性土壤(pH=8.07)连续三年的定位试验结果表明,生物质炭(pH=10.4)的施用并未显著提高土壤的pH,且仅在第一年表现出对小麦镉吸收的抑制效果。由此可见,石灰效应是生物质炭降低酸性土壤镉的生物有效性、抑制作物镉吸收的重要机制[44, 59-79]。
土壤的有机质含量对生物质炭的镉钝化效应也有一定的影响。通常,土壤中较高的有机质含量会促进重金属的钝化[79-82]。研究表明,生物质炭对可溶性有机质的吸附能够增加其表面官能团数量,从而增加生物质炭的吸附能力[83]。Chen等[25]的研究结果表明,当土壤有机质的含量较高时(> 15 g·kg-1),生物质炭对作物镉积累量的降低幅度较高。这可能是由于较高的土壤有机质含量可以在增加生物质炭表面官能团数量的同时提供更多的镉结合位点。此外,相关研究还表明,土壤中的无机、有机组分以及微生物可以与生物质炭相互作用,在生物质炭表面形成有机-矿质-生物覆膜,增加生物质炭表面官能团数量[86-87],这也可能会进一步促进生物质炭对镉的钝化。
2.3 管理方式除生物质炭制备条件及自身性质外,生物质炭的施用量以及施用深度也会影响生物质炭对土壤中镉的钝化效果。在10~40 t·hm-2施用量范围内,生物质炭对土壤中镉的钝化效应随着施用量的增加而提高[49-51, 77-78]。据统计,实际应用过程中生物质炭一般通过表层撒施、机械点施等方式将生物质炭翻耕于15 cm土层内,但也有至25 cm甚至更深的土层[79]。研究显示, 增加施用深度可显著提高作物产量,但作物对镉的积累量也有提高[52, 79]。这可能是由于深耕使生物质炭改善了作物根系区域的pH环境以及养分条件,促进了作物的生长,但由深耕带来的重金属下移也会增加作物根部对重金属的吸收[79, 86]。
3 结语目前为止,生物质炭对土壤镉钝化机制的认识还未深入到纳米或分子的水平,特别是关于生物质炭施用下土壤中矿物与有机质的相互作用的影响研究不多,这也是不同土壤治理效果产生较大差异的原因。另外,大量研究都提示,并不是所有生物质炭对镉都有显著的钝化效果,因此从生物质炭材料学角度加强性质与钝化功效研究,筛选最优材料或最优工艺在环境修复产业上显得尤为重要。最后,生物质炭镉污染治理技术还需要更多的大田定位长期试验研究,从而为生物质炭污染治理的生态系统服务功能和人类福祉的改善提供依据。
| [1] |
Tan Z X, Wang Y H, Kasiuliene A, et al. Cadmium removal potential by rice straw-derived magnetic biochar[J]. Clean Technologies and Environmental Policy, 2017, 19(3): 761-774. DOI:10.1007/s10098-016-1264-2 |
| [2] |
Melo L C A, Puga A P, Coscione A R, et al. Sorption and desorption of cadmium and zinc in two tropical soils amended with sugarcane-strawderived biochar[J]. Journal of Soils and Sediments, 2016, 16(1): 226-234. DOI:10.1007/s11368-015-1199-y |
| [3] |
Mench M, Lepp N, Bert V, et al. Successes and limitations of phytotechnologies at field scale:Outcomes, assessment and outlook from COST Action 859[J]. Journal of Soils and Sediments, 2010, 10(6): 1039-1070. DOI:10.1007/s11368-010-0190-x |
| [4] |
Sud D, Mahajan G, Kaur M P. Agricultural waste material as potential adsorbent for sequestering heavy metal ions from aqueous solutions:A review[J]. Bioresource Technology, 2008, 99(14): 6017-6027. DOI:10.1016/j.biortech.2007.11.064 |
| [5] |
Belhalfaoui B, Aziz A, Elandaloussi E H, et al. Succinate-bonded cellulose:A regenerable and powerful sorbent for cadmium-removal from spiked high-hardness groundwater[J]. Journal of Hazardous Materials, 2009, 169(1/2/3): 831-837. |
| [6] |
宋波, 曾炜铨. 土壤有机质对镉污染土壤修复的影响[J]. 土壤通报, 2015, 46(4): 1018-1024. SONG Bo, ZENG Wei-quan. Eeffects of organic matteronthe remediation of cadmium-contaminated soil:A review[J]. Chinese Journal of Soil Science, 2015, 46(4): 1018-1024. |
| [7] |
环境保护部, 国土资源部. 全国土壤污染状况调查公报[R]. 北京, 2014. The Ministry of Environmental Protection, The Ministry of Land and Resources. National soil contamination survey report[R]. Beijing, 2014. |
| [8] |
Liu X J, Tian G J, Jiang D, et al. Cadmium(Cd)distribution and contamination in Chinese paddy soils on national scale[J]. Environmental Science and Pollution Research, 2016, 23(18): 17941-17952. DOI:10.1007/s11356-016-6968-7 |
| [9] |
甄艳红, 成艳君, 潘根兴, 等. 中国部分市售大米中Cd、Zn、Se的含量及其食物安全评价[J]. 安全与环境学报, 2008, 8(1): 119-122. ZHEN Yan-hong, CHENG Yan-jun, PAN Gen-xing, et al. Cd, Zn and Se content of the polished rice samples from some Chinese open markets and their relevance to food safety[J]. Journal of Safety and Environment, 2008, 8(1): 119-122. |
| [10] |
张良运, 李恋卿, 潘根兴. 南方典型产地大米Cd、Zn、Se含量变异及其健康风险探讨[J]. 环境科学, 2009, 30(9): 2792-2797. ZHANG Liang-yun, LI Lian-qing, PAN Gen-xing. Variation of Cd, Zn and Se contents of polished rice and thepotential health risk for subsistence-diet farmers from typical areas of South China[J]. Environmental Science, 2009, 30(9): 2792-2797. |
| [11] |
Peng X, Deng Y G, Peng Y, et al. Effects of biochar addition on toxic element concentrations in plants:A meta-analysis[J]. Science of the Total Environment, 2018, 616: 970-977. |
| [12] |
Sohi S P, Krull E, Lopez-Capel E, et al. A review of biochar and its use and function in soil[J]. Advances in Agronomy, 2010, 105: 47-82. DOI:10.1016/S0065-2113(10)05002-9 |
| [13] |
Li H B, Dong X L, daSilva EB, et al. Mechanisms of metal sorption by biochars:Biochar characteristics and modifications[J]. Chemosphere, 2017, 178: 466-478. DOI:10.1016/j.chemosphere.2017.03.072 |
| [14] |
Lehmann J. Bio-energy in the black[J]. Frontiers in Ecology and the Environment, 2007, 5(7): 381-387. DOI:10.1890/1540-9295(2007)5[381:BITB]2.0.CO;2 |
| [15] |
Bird M I, AscoughP L, Young I M, et al. X-ray microtomographic imaging of charcoal[J]. Journal of Archaeological Science, 2008, 35(10): 2698-2706. DOI:10.1016/j.jas.2008.04.018 |
| [16] |
Kimetu J M, Lehmann J, Ngoze S O, et al. Reversibility of soil productivity decline with organic matter of differing quality along a degradation gradient[J]. Ecosystems, 2008, 11(5): 726-739. DOI:10.1007/s10021-008-9154-z |
| [17] |
Nguyen B T, Lehmann J, Kinyangi J, et al. Long-term black carbon dynamics in cultivated soil[J]. Biogeochemistry, 2009, 92(1/2): 163-176. |
| [18] |
IBI. Standardized product definition and product testing guidelines for biochar that is used in soil[EB/OL]. [2015-11-23]. http://www.biochar-international.org/characterizationstandard.
|
| [19] |
Mohan D, Pittman C U, Bricka M, et al. Sorption of arsenic, cadmium, and lead by chars produced from fast pyrolysis of wood and bark during bio-oil production[J]. Journal of Colloid and Interface Science, 2007, 310(1): 57-73. DOI:10.1016/j.jcis.2007.01.020 |
| [20] |
Cao X D, Ma L, Gao B, et al. Dairy-manure derived biochar effectively sorbs lead and atrazine[J]. Environmental Science & Technology, 2009, 43(9): 3285-3291. |
| [21] |
Park J H, Choppala G K, Bolan N S, et al. Biochar reduces t h e bioavailability and phytotoxicity of heavy metals[J]. Plant and Soil, 2011, 348(1/2): 439-451. |
| [22] |
Pan G X, Li L Q. The remediation technology for metal contaminated soil[J]. China Rural Science and Technol, 2013, 40-41. |
| [23] |
黄益宗, 郝晓伟, 雷鸣, 等. 重金属污染土壤修复技术及其修复实践[J]. 农业环境科学学报, 2013, 32(3): 409-417. HUANG Yi-zong, HAO Xiao-wei, LEI Ming, et al. The remediation technology and remediation practice of heavy metals-contaminated soil[J]. Journal of Agro-Environment Science, 2013, 32(3): 409-417. |
| [24] |
Wang M M, Zhu Y, Cheng L R, et al. Review on utilization of biochar for metal-contaminated soil and sediment remediation[J]. Journal of Environmental Sciences, 2018, 63(1): 156-173. |
| [25] |
Chen D, Liu X Y, Bian R J, et al. Effects of biochar on availability and plant uptake of heavy metals:A meta-analysis[J]. Journal of Environmental Management, 2018, 222: 76-85. |
| [26] |
Liu X L, Zeng Z X, Tie B Q, et al. Effects of biochar and lime application on soluble Cd, Pb, as release and non-point loads of rice agroecosystem by in situ field experiment, central Hunan Province mining area[C]//Proceedings of the 35th IAHR World Congress, Chengdu, 2013: 2264-2272.
|
| [27] |
He T Y, Meng J, Chen W F, et al. Effects of biochar on cadmium accumulation in rice and cadmium fractions of soil:A three-year pot experiment[J]. Bioresources, 2016, 12(1): 622-642. |
| [28] |
Abbas T, Rizwan M, Ali S, et al. Effect of biochar on cadmium bioavailability and uptake in wheat(Triticum aestivum L.)grown in a soil with aged contamination[J]. Ecotoxicology and Environmental Safety, 2017, 140: 37-47. DOI:10.1016/j.ecoenv.2017.02.028 |
| [29] |
Ahmad M, Rajapaksha A U, Lim J E, et al. Biochar as a sorbent for contaminant management in soil and water:A review[J]. Chemosphere, 2014, 99(3): 19-33. |
| [30] |
Wu W X, Yang M, Feng Q B, et al. Chemical characterization of rice straw-derived biochar for soil amendment[J]. Biomass and Bioenergy, 2012, 47(4): 268-276. |
| [31] |
Paz-Ferreiro J, Lu H, Fu S, et al. Use of phytoremediation and biochar to remediate heavy metal polluted soils:A review[J]. Solid Earth Discussions, 2014, 5(2): 2155-2179. |
| [32] |
Lu H L, Zhang W H, Yang Y X, et al. Relative distribution of Pb2+ sorption mechanisms by sludge-derived biochar[J]. Water Research, 2012, 46(3): 854-862. DOI:10.1016/j.watres.2011.11.058 |
| [33] |
Cantrell K B, Hunt P G, Uchimiya M, et al. Impact of pyrolysis temperature and manure source on physicochemical characteristics of biochar[J]. Bioresource Technol, 2012, 107(2): 419-428. |
| [34] |
Gul S, Whalen J K, Thomas B W, et al. Physico-chemical properties and microbial responses in biochar-amended soils:Mechanisms and future directions[J]. Agriculture Ecosystems & Environment, 2015, 206: 46-59. |
| [35] |
Yousaf B, Liu G J, Wang R W, et al. Investigating the potential influence of biocharand traditional organic amendments on the bioavailability and transfer of Cd in thesoil plant system[J]. Environmental Earth Sciences, 2016, 75(5): 374. DOI:10.1007/s12665-016-5285-2 |
| [36] |
Namgay T, Singh B, Singh B P, et al. Influence of biochar application to soil on the availability of As, Cd, Cu, Pb, and Zn to maize(Zea mays L.)[J]. Australian Journal of Soil Research, 2010, 48(6/7): 638-647. |
| [37] |
Zhang F, Wang X, Yin D X, et al. Efficiency and mechanisms of Cd removal from aqueous solution by biochar derived from water hyacinth (Eichornia crassipes)[J]. Journal of Environmental Management, 2015, 153: 68-73. |
| [38] |
Harvey O R, Herbert B E, Rhue R D, et al. Metal interactions at the biochar-water interface:Energetics and structure-sorption relationships elucidated by flow adsorption microcalorimetry[J]. Environmental Science & Technology, 2011, 45(13): 5550-5556. |
| [39] |
Xu X Y, Cao X D, Zhao L. Comparison of rice husk-and dairy manure-derived biochars for simultaneously removing heavy metals from aqueous solutions:Role of mineral components in biochars[J]. Chemosphere, 2013, 92(8): 955-961. DOI:10.1016/j.chemosphere.2013.03.009 |
| [40] |
Mohamed I, Zhang G S, Li Z G, et al. Ecological restoration of an acidic Cd contaminated soil using bamboo biochar application[J]. Ecological Engineering, 2015, 84: 67-76. DOI:10.1016/j.ecoleng.2015.07.009 |
| [41] |
Joseph S D, Camps-Arbestain M, Lin Y, et al. An investigation into the reactions of biochar in soil[J]. Soil Research, 2010, 48(6/7): 501-515. |
| [42] |
Major J, Steiner C, Downie A, et al. Biochar effects on nutrient leaching[M]//Biochar for environmental management. London: Earthscan. 2009: 271-288.
|
| [43] |
Shinogi Y, Yoshida H, Koizumi T, et al. Basic characteristics of lowtemperature carbon products from waste sludge[J]. Advances in Environmental Research, 2003, 7(3): 661-665. DOI:10.1016/S1093-0191(02)00040-0 |
| [44] |
Rizwan M, Ali S, Qayyum M F, et al. Mechanisms of biochar-mediated alleviation of toxicity of trace elements in plants:A critical review[J]. Environmental Science and Pollution Research, 2016, 23(3): 2230-2248. DOI:10.1007/s11356-015-5697-7 |
| [45] |
Shi R.Y, Hong Z N, Li J Y, et al. Mechanisms for increasing the pH buffering capacity of an acidic ultisol by crop residue-derived biochars[J]. Journal of Agricultural and Food Chemistry, 2017, 65(37): 8111-8119. DOI:10.1021/acs.jafc.7b02266 |
| [46] |
Rees F, Germain C, Sterckeman T, et al. Plant growth and metal uptake by a non-hyperaccumulating species(Lolium perenne)and a CdZn hyperaccumulator(Noccaea caerulescens) in contaminated soils amended with biochar[J]. Plant and Soil, 2015, 395(1/2): 57-73. |
| [47] |
Trakal L, Bingol D, Pohorely M, et al. Geochemical and spectroscopic investigations of Cd and Pb sorption mechanisms on contrasting biochars:Engineering implications[J]. Bioresource Technology, 2014, 171: 442-451. DOI:10.1016/j.biortech.2014.08.108 |
| [48] |
Lahori A H, Guo Z Y, Zhang Z Q, et al. Use of biochar as an amendment for remediation of heavy metal-contaminated soils:Prospects and challenges[J]. Pedosphere, 2017, 27(6): 991-1014. DOI:10.1016/S1002-0160(17)60490-9 |
| [49] |
Bian R J, Joseph S, Cui L Q, et al. A three-year experiment confirms continuous immobilization of cadmium and lead in contaminated paddy field with biochar amendment[J]. Journal of Hazardous Materials, 2014, 272(4): 121-128. |
| [50] |
Cui L Q, Pan G X, Li L Q, et al. The reduction of wheat Cd uptake in contaminated soil via biochar amendment:A two-year field experiment[J]. Bioresources, 2012, 7(4): 5666-5676. |
| [51] |
Cui L Q, Li L Q, Zhang A F, et al. Biochar amendment greatly reduces rice Cd uptake in a contaminated paddy soil:A two-year field experiment[J]. Bioresources, 2011, 6(3): 2605-2618. |
| [52] |
Xu M, Wu J, Luo L, et al. The factors affecting biochar application in restoring heavy metal-polluted soil and its potential applications[J]. Chemistry and Ecology, 2017, 34(2): 177-197. |
| [53] |
Chaney R L, Reeves P G, Ryan J A, et al. An improved understanding of soil Cd risk to humans and low-cost methods to phytoextract Cd from contaminated soils to prevent soil Cd risks[J]. Biometals, 2004, 17(5): 549-553. DOI:10.1023/B:BIOM.0000045737.85738.cf |
| [54] |
史静, 潘根兴, 张乃明. 镉胁迫对不同杂交水稻品种Cd、Zn吸收与积累的影响[J]. 环境科学学报, 2013, 33(10): 2904-2910. SHI Jing, PAN Gen-xing, ZHANG Nai -ming. Effect of cadmium stress on Cd and Zn uptake and accumulation of different cultivars of hybrid rice[J]. Acta Scientiae Circumstantiae, 2013, 33(10): 2904-2910. |
| [55] |
Hassan M J, Zhu Z, Ahmad B, et al. Influence of cadmium toxicity on rice genotypes as affected by zinc, sulfur and nitrogen fertilizers[J]. Caspian Journal of Environmental Sciences, 2006, 4(1): 1-8. |
| [56] |
Chen D, Guo H, Li R Y, et al. Low uptake affinity cultivars with biochar to tackle Cd-tainted rice:A field study over four rice seasons in Hunan, China[J]. Science of the Total Environment, 2016, 541: 1489-1498. DOI:10.1016/j.scitotenv.2015.10.052 |
| [57] |
Rizwan M, Ali S, Adrees M, et al. Cadmium stress in rice:Toxic effects, tolerance mechanisms, and management:A critical review[J]. Environmental Science and Pollution Research International, 2016, 23(18): 17859-17879. DOI:10.1007/s11356-016-6436-4 |
| [58] |
Rizwan M, Ali S, Abbas T, et al. Cadmium minimization in wheat:A critical review[J]. Ecotoxicol Environ Saf, 2016, 130: 43-53. DOI:10.1016/j.ecoenv.2016.04.001 |
| [59] |
Kookana R S, Sarmah A K, Van Z L, et al. Biochar application to soil: Agronomic and environmental benefits and uninteneed consequences[M]//Sparks D L. Advances in Agronomy, Vol 112. 2011, 112: 103-143.
|
| [60] |
Yao F X, Arbestain M C, Virgel S, et al. Simulated geochemical weathering of a mineral ash-rich biochar in a modified Soxhlet reactor[J]. Chemosphere, 2010, 80(7): 724-732. DOI:10.1016/j.chemosphere.2010.05.026 |
| [61] |
Singh B, Singh B P, Cowie A L, et al. Characterisation and evaluation of biochars for their application as a soil amendment[J]. Soil Research, 2010, 48(6/7): 516-525. |
| [62] |
Kloss S, Zehetner F, Dellantonio A, et al. Characterization of slow pyrolysis biochars:Effects of feedstocks and pyrolysis temperature on biochar properties[J]. Journal of Environmental Quality, 2012, 41(4): 990-1000. DOI:10.2134/jeq2011.0070 |
| [63] |
Lehmann J, Rillig M C, Thies J, et al. Biochar effects on soil biota-A review[J]. Soil Biology & Biochemistry, 2011, 43(9): 1812-1836. |
| [64] |
Kibria M G, Osman K T, Ahmed M J. Cadmium and lead uptake by radish(Raphanus sativus L.)grown in three different textured soils[J]. Soil and Environment, 2007, 26(2): 106-114. |
| [65] |
Rafiq M T, Aziz R, Yang X, et al. Cadmium phytoavailability to rice (Oryza sativa L.)grown in representative Chinese soils. A model to improve soil environmental quality guidelines for food safety[J]. Ecotoxicol Environ Saf, 2014, 103(1): 101-107. |
| [66] |
Aziz R, Rafiq M T, Li T Q, et al. Uptake of cadmium by rice grown on contaminated soils and its bioavailability/toxicity in human cell lines (Caco-2/HL-7702)[J]. Journal of Agricultural and Food Chemistry, 2015, 63(13): 3599-3608. DOI:10.1021/jf505557g |
| [67] |
Zhang X K, Wang H L, He L Z, et al. Using biochar for remediation of soils contaminated with heavy metals and organic pollutants[J]. Environmental Science and Pollution Research, 2013, 20(12): 8472-8483. DOI:10.1007/s11356-013-1659-0 |
| [68] |
Najafi S, Jalali M. Effect of heavy metals on pH buffering capacity and solubility of Ca, Mg, K, and P in non-spiked and heavy metal-spiked soils[J]. Environmental Monitoring and Assessment, 2016, 188(6): 1-11. |
| [69] |
Uchimiya M, Chang S C, Klasson K T. Screening biochars for heavy metal retention in soil:Role of oxygen functional groups[J]. Journal of Hazardous Materials, 2011, 190(1/3): 432-441. |
| [70] |
Caporale A G, Violante A. Chemical processes affecting the mobility of heavy metals and metalloids in soil environments[J]. Curr Pollut Rep, 2016, 2(1): 15-27. DOI:10.1007/s40726-015-0024-y |
| [71] |
Gustafsson J P. Arsenate adsorption to soils:Modelling the competition from humic substances[J]. Geoderma, 2006, 136(1/2): 320-330. |
| [72] |
Jiang J, Xu R K, Jiang T Y, et al. Immobilization of Cu(Ⅱ), Pb(Ⅱ) and Cd(Ⅱ)by the addition of rice straw derived biochar to a simulat-ed polluted Ultisol[J]. Journal of Hazardous Materials, 2012, 229-230: 145-150. DOI:10.1016/j.jhazmat.2012.05.086 |
| [73] |
Houben D, Evrard L, Sonnet P. Mobility, bioavailability and pH-dependent leaching of cadmium, zinc and lead in a contaminated soil amended with biochar[J]. Chemosphere, 2013, 92(11): 1450-1457. DOI:10.1016/j.chemosphere.2013.03.055 |
| [74] |
Rees F, Simonnot M O, Morel J L. Short-term effects of biochar on soil heavy metal mobility are controlled by intra-particle diffusion and soil pH increase[J]. European Journal of Soil Science, 2014, 65(1): 149-161. DOI:10.1111/ejss.12107 |
| [75] |
Qi F J, Dong Z M, Lamb D, et al. Effects of acidic and neutral biochars on properties and cadmium retention of soils[J]. Chemosphere, 2017, 180: 564-573. DOI:10.1016/j.chemosphere.2017.04.014 |
| [76] |
Rees F, Sterckeman T, Morel J L. Root development of non-accumulating and hyperaccumulating plants in metal-contaminated soils amended with biochar[J]. Chemosphere, 2016, 142: 48-55. DOI:10.1016/j.chemosphere.2015.03.068 |
| [77] |
Bian R J. Effect of municipal biowaste biochar on greenhouse gas emissions and metal bioaccumulation in a slightly acidic clay rice paddy[J]. Bioresources, 2013, 9(1): 685-703. |
| [78] |
Sui F F, Zuo J, Chen D, et al. Biochar effects on uptake of cadmium and lead by wheat in relation to annual precipitation:A 3-year field study[J]. Environmental Science and Pollution Research, 2018, 25(4): 3368-3377. DOI:10.1007/s11356-017-0652-4 |
| [79] |
O'Connor D, Peng T Y, Zhang J L, et al. Biochar application for the remediation of heavy metal polluted land:A review of in situ field trials[J]. Science of the Total Environment, 2018, 619-620: 815-826. DOI:10.1016/j.scitotenv.2017.11.132 |
| [80] |
Xu P, Sun C X, Ye X Z, et al. The effect of biochar and crop straws on heavy metal bioavailability and plant accumulation in a Cd and Pb polluted soil[J]. Ecotoxicology and Environmental Safety, 2016, 132: 94-100. DOI:10.1016/j.ecoenv.2016.05.031 |
| [81] |
Jin S C, Park S H, Jung S C, et al. Production and utilization of biochar:A review[J]. Journal of Industrialand Engineering Chemistry, 2016, 40: 1-15. DOI:10.1016/j.jiec.2016.06.002 |
| [82] |
Venegas A, Rigol A, Vidal M. Changes in heavy metal extractability from contaminated soils remediated with organic waste or biochar[J]. Geoderma, 2016, 279: 132-140. DOI:10.1016/j.geoderma.2016.06.010 |
| [83] |
Heitkötter J, Marschner B. Interactive effects of biochar ageing in soils related to feedstock, pyrolysis temperature, and historic charcoal production[J]. Geoderma, 2015, 17: 56-64. |
| [84] |
Archanjo B S, Mendoza M E, Albu M, et al. Nanoscale analyses of the surface structure and composition of biochars extracted from field trials or after co-composting using advanced analytical electron microscopy[J]. Geoderma, 2017, 294: 70-79. DOI:10.1016/j.geoderma.2017.01.037 |
| [85] |
Hagemann N, Joseph S, Schmidt H P, et al. Organic coating on biochar explains its nutrient retention and stimulation of soil fertility[J]. Nature Communications, 2017, 8(1): 1089. DOI:10.1038/s41467-017-01123-0 |
| [86] |
Duering R A, Hoss T, Gaeth S. Sorption and bioavailability of heavy metals in long-term differently tilled soils amended with organic wastes[J]. Science of the Total Environment, 2003, 313(1/3): 227-234. |
 2018, Vol. 37
2018, Vol. 37